Max Planck researchers identify epithelial stem cells that control thymus development at different stages of life
Many immune cells, which are vital to our immune system, develop early in life and into adolescence in a small organ right next to our heart: the thymus. However, with age, the thymus shrinks and its function and thus the number of powerful immune cells decreases. Freiburg and Würzburg’s Max Planck research groups have now identified the processes that control the development and structure of thymus tissue during life. They also came across potential therapeutic approaches to reverse age-related thymus shrinkage and to treat autoimmune diseases.
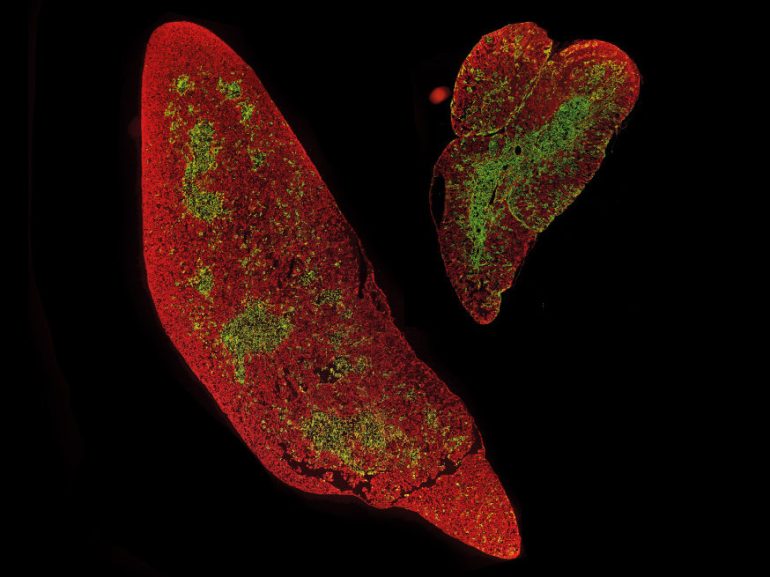
© MPI for Immunobiology and Epigenetics, Boehm
The tissue structure of the excised thymus organ does not differ from that of an unstimulated organ. As a sign of a well-functioning thymus, the edge region (red) and the inner region (green) are sharply separated from each other.
© MPI for Immunobiology and Epigenetics, Boehm
The thymus is a central organ of the immune system. This is where T-cells are made, which, as killer cells, recognize and destroy virus-infected or degenerated cells, as well as so-called helper T-cells, which help the body make antibodies. .
Over the past few decades, Thomas Boehm’s research group at the Max Planck Institute of Immunobiology and Epigenetics in Freiburg has been able to identify the genetic switch required in the thymus for T cell maturation. So-called thymic epithelial cells, which attract T-cell precursors and bring them to maturity, are essential here. T-cells learn to differentiate diseased cells from healthy and foreign cells. With it you can detect and eliminate unwanted formations and prevent autoimmune diseases. Previous work in the laboratory of Thomas Boehm showed that the two main forms of the thymic epithelium arise from biphasic progenitor cells. Until now, however, it has not been clear whether there is more than one type of precursor and how many of the subtypes differ in precursors.
Lineage analysis identifies progenitor cells of the thymic epithelium
with the support of Dominic Green’s lab (formerly the Max Planck Institute for Immunobiology and Epigenetics, now the Max Planck Research Group at the University of Würzburg), experts in molecular single cell analysis, have now molecularly recorded a surprisingly large diversity of thymic epithelial cells. Algorithms developed in Grun’s laboratory for precise description of differences in gene activity of individual cells made it possible to identify cells with potential precursor activity.
In a second phase, these predictions were tested experimentally by researchers using a “barcoding” system developed in Thomas Boehm’s lab using CRISPR gene scissors. The barcoding process makes it possible to provide progenitor cells with a molecular signature that is carried by all cells that descend from progenitors. This makes it possible to obtain a family tree of epithelial cells.
Anja Nusser from the Boehm laboratory and Sagar from the Grun laboratory have also jointly developed a method that combines information from the family tree with the molecular characteristics of individual cells. This made it possible for the first time to study the development of the thymic epithelium at different ages. This detail is of particular interest to immunologists because the thymus undergoes major changes throughout life. Early stages of development lead to rapid organ growth and massive T cell production, whereas old age leads to a gradual loss of functional thymus epithelial cells and therefore reduced T cell production. Age-related loss of function is associated with a diminished immune system.
The order of progenitor cells determines the structure of thymic tissue
© MPI for Immunobiology and Epigenetics, Boehm
Mass enlargement of the thymus organ stimulated even in young rats, which largely persists into old age.
© MPI for Immunobiology and Epigenetics, Boehm
In their analysis, the researchers were able to identify two diploid precursor populations of the thymus epithelium. An “early” progenitor population assumes a key role in thymus formation during the embryonic stage and in the juvenile organism, while a later “postnatal” progenitor population largely determines further thymus formation in adulthood. The composition of the thymus epithelium is modified differently by the temporal sequence of the progenitor population.
At early time points, mainly cortical thymic epithelial cells are formed, which mainly contribute to the production of T cells, while at later time points, the main production occurs on medullary thymic epithelial cells, which ensures that no self-reactive T cells are released. Enter the body through the thymus and thus contribute significantly to protection against autoimmunity.
New approaches to enhance thymus function
The efficient combination of transgenic animal models from the Boehm laboratory with the most modern methods of single cell characterization in the Grun working group enabled researchers to examine the effect of a method for the proliferation of epithelial cells, which has been known for years. Thymus. This was of particular importance to determine whether initial stimulation of the thymus with a particular growth factor leads to an undesirably rapid consumption of stem cells and thus early shrinkage of the thymus.
Team statistics show that is not the case. “The thymus we stimulated in an older mouse is still larger than that of an unstimulated young mouse. The tissue structure of the stimulated thymus also reflects the normal structure of the cortical zones and medullary regions inside the organ,” says Max Planck, director of With these results, the Freiburg researchers have laid the foundation for developing new therapeutic approaches to reverse age-related thymus shrinkage and treat T-cell-dependent autoimmune diseases, says Thomas Boehm.
TB/MR

Web guru. Amateur thinker. Unapologetic problem solver. Zombie expert. Hipster-friendly travel geek. Social mediaholic.