DHe said that the European Union Commission and the Italian Government are proud to have 250,000 doses of the Astra-Zeneca vaccine in the European Union. On the other hand, the Australian government is no doubt – and there is also increasing criticism in the European Union of the export ban, with which the government in Rome, with the approval of Brussels, prevents vaccine bottles from being transported to Australia in Italy.
Christoph Hein
Business correspondent for South Asia / Pacific based in Singapore.
Unfortunately, it is not surprising at these times that Australia’s Treasury Secretary Michael Birmingham said that some countries would “tear down the rule book”. The chairman of the trade committee in the European Parliament, Bernad Lang (SPD), criticized the Italian decision as opening a “Pandora’s box”. It threatens to lead a global battle for vaccines, in which the European Union may have no interest.
The export ban emerged a week ago at a video conference of heads of state and government. Italy’s new Prime Minister Mario Draghi, who wants to “militarize” his vaccination campaign, demanded that the EU should generally ensure that vaccines produced or packaged in member states also remain in the EU . Drew did not announce a particular Italian decision.
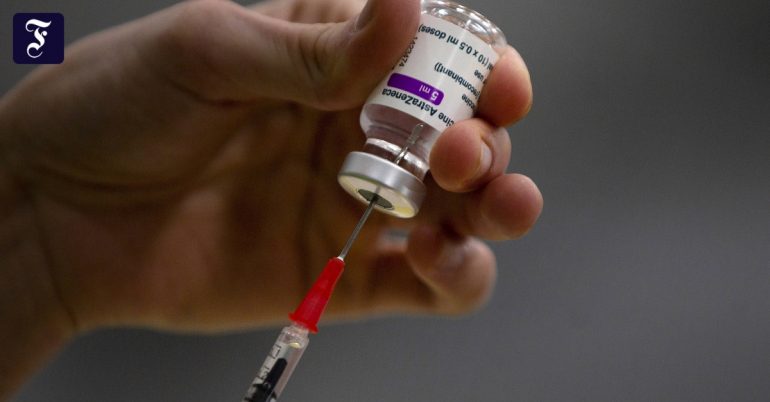
It fell after some time and was then approved by the European Union Commission on Monday – according to the rules. Affected is the vaccine that Agne-Zeneca (AZ) fills in Anagni in the south of Rome.
“Nuclear option” only
The export ban has sharply sparked controversy by the EU Commission over the general handling of vaccine shortages – more than expected about the “export mechanism” that the EU Commission introduced practically overnight at the end of January did. This action was a direct response to delivery failures that became apparent in AZ at the time.
In the words of Trade Commissioner Waldis Dombrovsky, the “mechanism” is supposed to include only a transparency register whereby manufacturers have to register exports. An export ban was not associated with it, the Latvian said at the time.
Elsewhere in the European Union authority, however, it was already noted in January that member states could stop exports “in rare cases” and “in emergencies”. But that is only the “nuclear option”. The “basic assumption” is that exports are possible. An emergency is clearly that export is at the expense of delivery to the European Union that a manufacturer has promised.
A lex as
This is why the agreed “mechanism” is also known as lex as. The European Union Commission is targeting the British-Swedish manufacturer as it became clear in January that it would not be able to meet the delivery volume specified in the distribution contract with the European Union in the first or second quarter. How the formula contained in the contract is to be interpreted, that AZ has to do the best possible (“best effort”) for delivery, is legally controversial and will only have to be clarified in court when the vaccine is lacking (expected Is) up.
The EU Commission suspects that the AZ vaccine, produced in the European Union, was exported to Great Britain in December at the expense of the European Union. AZ delivery commitments for the second quarter are no longer considered reliable among EU officials.
In contrast, the Commission emphasizes that exports will certainly be approved by companies keeping their delivery commitments to the European Union. In 150 cases, approval was granted. This also applies to Australia. For example, Biontech-Pfizer may export its vaccine to Australia, EU Ambassador to Canberra, Michael Palch, assured FAZ
Criticism not only from Australia
Apparently the disaster was already coming on the fifth continent. The Australian Chamber of Commerce called on the United Nations in mid-February to leave the “apparatus” with chambers in New Zealand, Canada, Chile, Korea, Brazil, Turkey and the United States. “We are deeply concerned that the move will set a precedent and encourage other countries to follow suit,” it reads.

Reader. Organizer. General creator. Zombie fanatic. Alcohol advocate. Food junkie. Bacon ninja.